

Expanded phase II B clinical trials of Cuba’s anti-COVID-19 candidate vaccine Soberana 02 with volunteers between 19 and 80 years of age, began at the 19 de Abril and No.1 Polyclinics, in the Havana municipalities of Plaza de la Revolución and La Lisa, respectively.
For the purpose of evaluating reactions, safety and immunogenicity of the candidate, created by the Finlay Vaccine Institute, volunteers will receive two doses of the drug, administered intramuscularly, and results will be compared to those from a placebo control group.

First steps for participating volunteers included signing an informed consent document and evaluation by medical staff.
The principal researcher leading this stage, Dr. María Eugenia Toledo, from the Pedro Kourí Institute of Tropical Medicine, explained that Soberana 02 is a conjugated vaccine, in which the virus antigen is chemically linked to the tetanus toxoid, and that after the results of this phase are evaluated, the vaccine will undergo a third phase to determine its clinical efficacy.
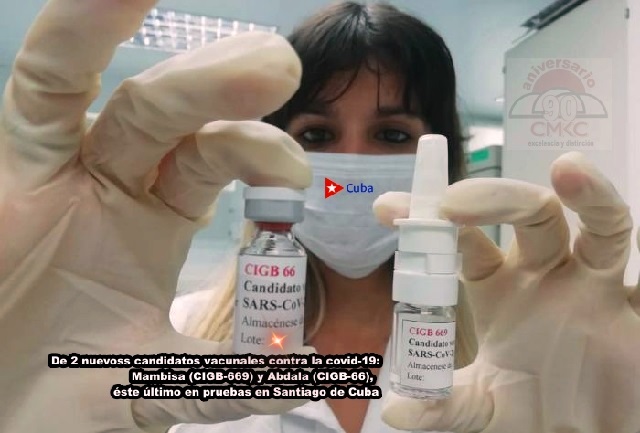
Since August, the Center for the State Control of Drugs, Equipment and Medical Devices has also authorized clinical trials of the anti-COVID-19 candidate vaccine Soberana 01, with which Cuba became the first country in Latin America and the Caribbean to develop a drug of this kind. At the beginning of November, Soberana 02 was authorized, as well as Mambisa (CIGB 669) and Abdala (CIGB 66), which are also being tested.
The polyclinic in Plaza de la Revolución was selected because the municipality is considered at high risk for transmission of the virus, and has participated in previous studies of interventions including administration of the VA-Mengoc-BC vaccine, to stimulate the residents’ immune response.

































































